No products in the cart.
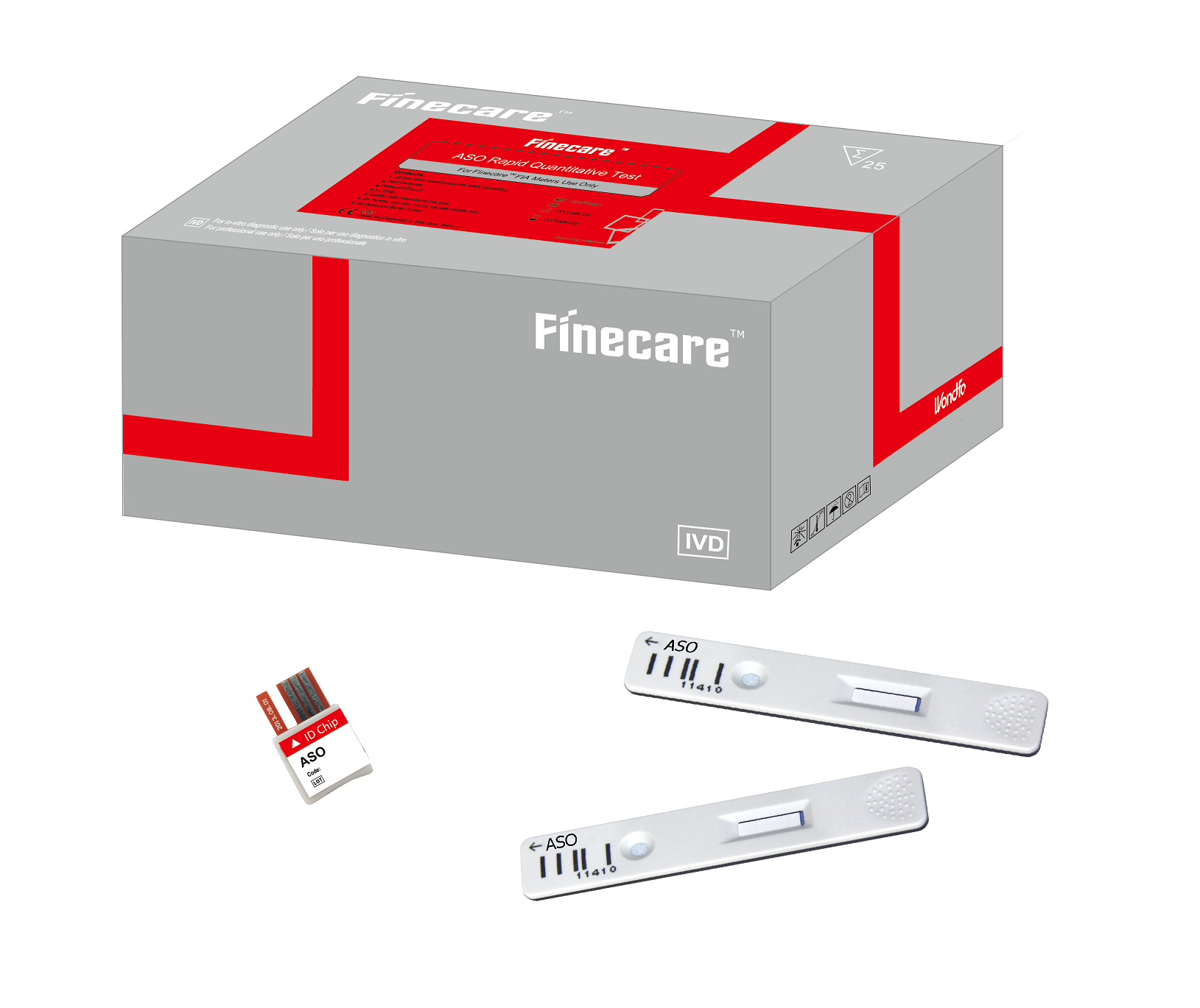
Finecare HbA1c Rapid Quantitative Test
4,750.00৳ Original price was: 4,750.00৳ .4,250.00৳ Current price is: 4,250.00৳ .
- Sample Type: Whole blood.
- Sample Volume: Requires 5 µL of the sample
- Reaction Time: Provides results in approximately 5 minutes.
- Measuring Range: Capable of measuring HbA1c concentrations between 4.0% and 14.5%.
- Certifications: CE, NGSP, and IFCC certified.
- USE for Wondfo Finecare
- Origin Korea
- Quantity 25 Test Device
Category POCT Test Kit
Tags Finecare, Finecare POCT Test, POCT Test Kit

01405100400
Free Delivery
For order. over 15,000 Taka
7 Days Return
If products have problems
Secure Payment
100% secure online payment.
Dedicated Support
Dedicated online and offline support
The Finecare™ HbA1c Rapid Quantitative Test is a fluorescence immunoassay designed for the quantitative determination of Hemoglobin A1c (HbA1c) levels in human whole blood. This test, used in conjunction with the Finecare™ FIA System, aids in monitoring long-term glycemic control in patients with diabetes mellitus.
Key Features:
- Sample Type: Whole blood.
- Sample Volume: Requires 5 µL of the sample
- Reaction Time: Provides results in approximately 5 minutes.
- Measuring Range: Capable of measuring HbA1c concentrations between 4.0% and 14.5%.
- Certifications: CE, NGSP, and IFCC certified.
Storage and Stability:
- Store the test kit at temperatures between 4℃ and 30℃ up to the expiration date printed on the package.
- If refrigerated, allow the test kit to reach room temperature (approximately 30 minutes) before testing.
- Use the test cartridge within one hour of opening the pouch.
Clinical Significance:
HbA1c is formed by the attachment of glucose to hemoglobin molecules in red blood cells and reflects the average blood glucose level over the past 2-3 months. Monitoring HbA1c levels is crucial for assessing long-term glycemic control in diabetic patients. Normal HbA1c levels in individuals without diabetes range from 4% to 5.9%. Patients with diabetes typically have HbA1c levels above 7%. Reducing HbA1c levels by 1% can decrease the risk of microvascular complications by 10%.
Performance Evaluation:
A study evaluating the Finecare™ HbA1c Rapid Quantitative Test demonstrated that it is a reliable and rapid assay (5 minutes) suitable for long-term monitoring of HbA1c in diabetic patients, particularly in small laboratory settings.
Usage Considerations:
- Ensure that the test cartridge, buffer, and ID chip lot numbers match before use.
- Allow the test kit to reach room temperature if previously refrigerated.
- Use the test cartridge within one hour of opening the pouch.
- Follow proper laboratory safety techniques when handling specimens and kit reagents.
Brand
Finecare
Wondfo
Be the first to review “Finecare HbA1c Rapid Quantitative Test” Cancel reply
Related Products
Related products
-
POCT Test Kit
IgE (Total) Rapid Test Device – Apoti POCT Analyzer
Rated 0 out of 55,000.00৳Original price was: 5,000.00৳ .4,250.00৳ Current price is: 4,250.00৳ . Add to cart -
POCT Test Kit
Troponin I Rapid Test Device – Apoti POCT Analyzer
Rated 0 out of 55,000.00৳Original price was: 5,000.00৳ .4,250.00৳ Current price is: 4,250.00৳ . Add to cart -
POCT Test Kit
Progesterone (Prog) Fast Test Kit for Hipro Palm
Rated 0 out of 5250.00৳Original price was: 250.00৳ .200.00৳ Current price is: 200.00৳ . Add to cart -
POCT Test Kit
HbA1c Rapid Test Device – Apoti POCT Analyzer
Rated 0 out of 54,800.00৳Original price was: 4,800.00৳ .4,500.00৳ Current price is: 4,500.00৳ . Add to cart
Our Best Selling Products
-
Mesh Nebulizer
Mesh Nebulizer – Portable Ultrasonic Inhaler Mesh Nebulizer
Rated 0 out of 52,750.00৳Original price was: 2,750.00৳ .1,800.00৳ Current price is: 1,800.00৳ . Add to cart -
Mesh Nebulizer
Mesh Nebulizer JSL-W301 – Portable Ultrasonic Inhaler Mesh Nebulizer
Rated 0 out of 52,500.00৳Original price was: 2,500.00৳ .1,500.00৳ Current price is: 1,500.00৳ . Add to cart -
Biochemistry Analyzer
Mindray BA-88A Semi-Auto Chemistry Analyzer
Rated 0 out of 5180,000.00৳Original price was: 180,000.00৳ .150,000.00৳ Current price is: 150,000.00৳ . Add to cart -
CLIA System
AutoLumo A1860 – Automatic Chemiluminescence Immunoassay System
Rated 0 out of 51,800,000.00৳Original price was: 1,800,000.00৳ .1,600,000.00৳ Current price is: 1,600,000.00৳ . Add to cart -
Microscope/Binocular
Cypress CM001 Cyanscope Microscope
Rated 0 out of 5140,000.00৳Original price was: 140,000.00৳ .110,000.00৳ Current price is: 110,000.00৳ . Add to cart -
Ultrasound Machine
Mindray DC-N3 Pro Ultrasound Machine
Rated 0 out of 51,350,000.00৳Original price was: 1,350,000.00৳ .1,150,000.00৳ Current price is: 1,150,000.00৳ . Add to cart -
Serology Reagent
S.Paratyphi-BH Antigen Serology Reagent – Human Biolin
Rated 0 out of 51,000.00৳Original price was: 1,000.00৳ .800.00৳ Current price is: 800.00৳ . Add to cart -
Patient Monitor
EMS-15000A Patient Monitor – Esonic
Rated 0 out of 570,000.00৳Original price was: 70,000.00৳ .65,000.00৳ Current price is: 65,000.00৳ . Add to cart














Reviews
There are no reviews yet.